13.4: Ways of Expressing Concentration - Chemistry LibreTexts
4.9 (635) · € 19.00 · En Stock
Different units are used to express the concentrations of a solution depending on the application. The concentration of a solution is the quantity of solute in a given quantity of solution. It can be …
Different units are used to express the concentrations of a solution depending on the application. The concentration of a solution is the quantity of solute in a given quantity of solution. It can be expressed in several ways.
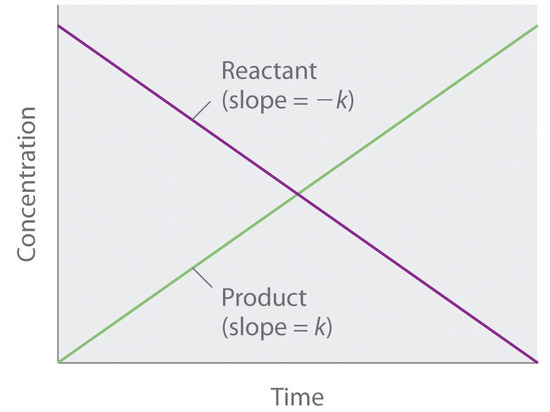
5.2: Methods of Determining Reaction Order - Chemistry LibreTexts

13.4 Expressing Concentration

15.3 Relative Strengths of Acids and Bases – Chemistry Fundamentals

Molar Mass Lab Report
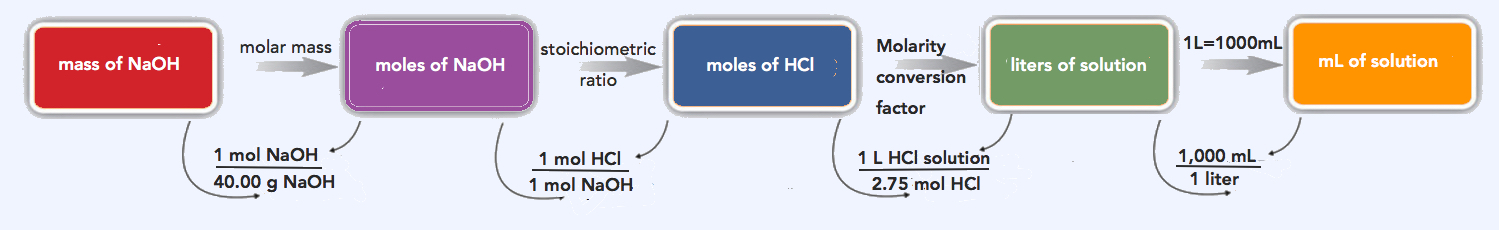
6.3: Concentration - Biology LibreTexts

13.4: The Law of Chemical Equilibrium: Equilibrium Constant in Other Sections, PDF, Acid
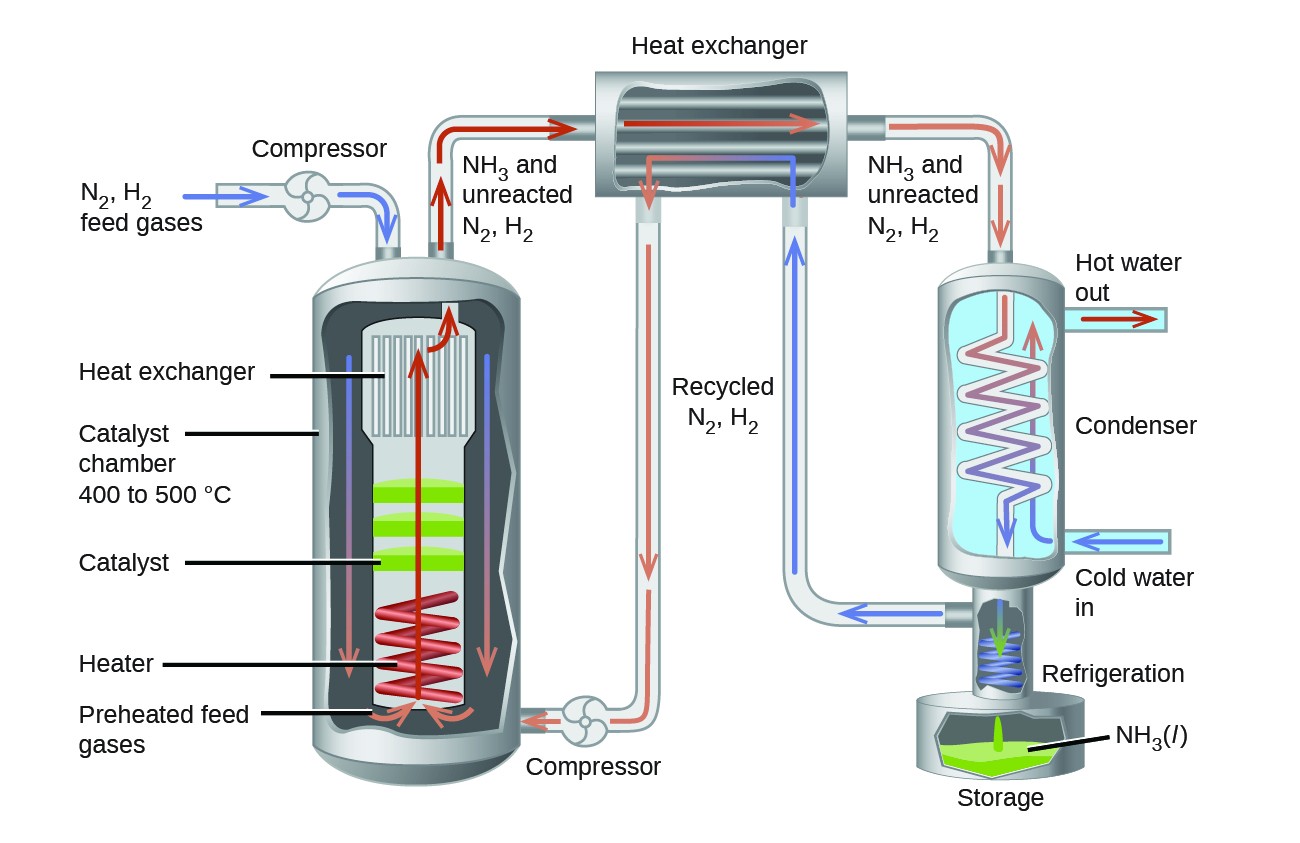
14.3 Shifting Equilibria: Le Châtelier's Principle – Chemistry Fundamentals

16.2 Lewis Acids and Bases – Chemistry Fundamentals

Indoor acids and bases - Nazaroff - 2020 - Indoor Air - Wiley Online Library

8.6 Other Units for Solution Concentrations – Chemistry Fundamentals
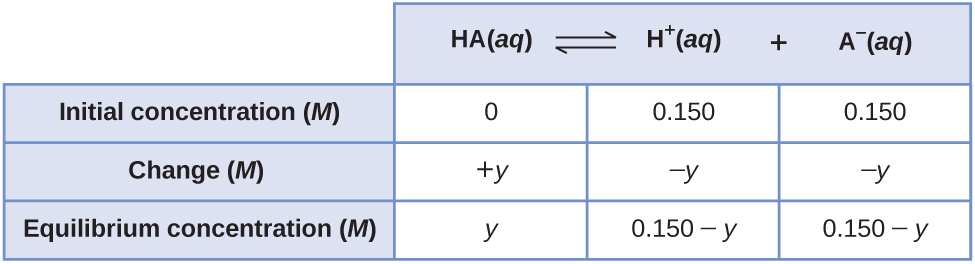
13.4: Equilibrium Calculations - Chemistry LibreTexts












